
Uncaking Colorless Crystals Industrial Grade Na2s2o3 5H2O Sodium Thiosulfate - China Sodium Thiosulfate Anhydrous and Sodium Hyposulfite

10102-17-7, 248.19, Sodium Thiosulfate, Pentahydrate, Crystal, USP, EP, BP, JP - 6WYT7|SO210-500GM - Grainger
What mass of Na2S2O3.5H2O is needed to make 500 cm^3 of 0.200 N solution for the reaction : - Sarthaks eConnect | Largest Online Education Community

Spectrum S1502-500GM (SPFDB) Sodium Thiosulfate, Pentahydrate, Crystal, FCC, Na2S2O3.5H2O: Amazon.com: Industrial & Scientific

Low Price Na2s2o3 5h2o Sodium Thiosulfate with Good Quality-Sodium thiosulfate-Tianjin Chengyuan Chemical Co., Ltd-Chengyuan

Spectrum S1502-500GM (SPFDB) Sodium Thiosulfate, Pentahydrate, Crystal, FCC, Na2S2O3.5H2O: Amazon.com: Industrial & Scientific
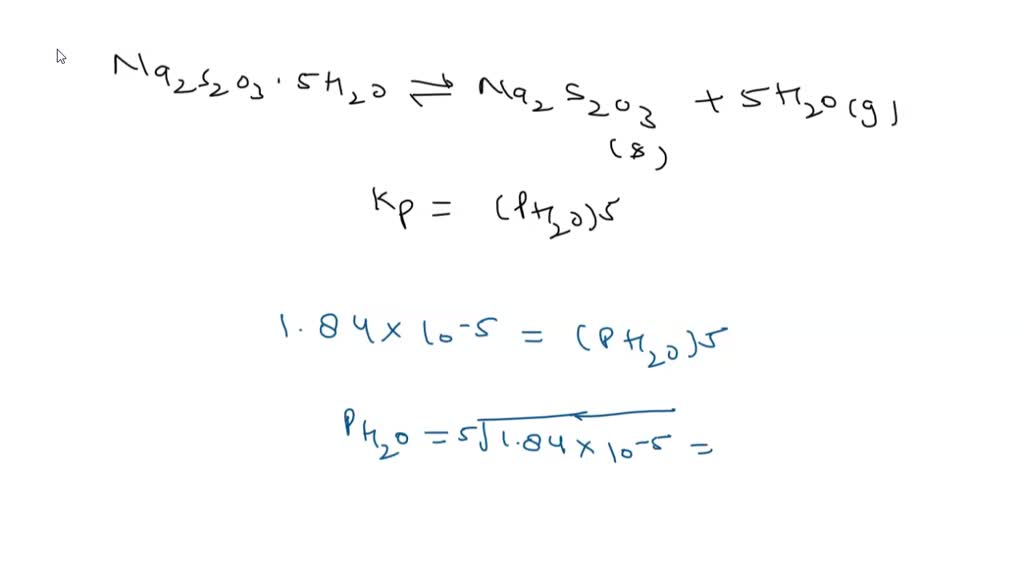
SOLVED: Sodium thiosulfate pentahydrate (FM 248.18 g ∙ mol-1) loses water when it is heated in an oven: Na2S2O3∙5H2O(s) ⇌ Na2S2O3(s) + 5H2O(g). ΔH° and ΔS° for this reaction at 25°C are







![Sodium Thiosulfate Pentahydrate [Na2O3S2.5H2O] [CAS_10102-17-7] Photo – Wintersun Chemical Sodium Thiosulfate Pentahydrate [Na2O3S2.5H2O] [CAS_10102-17-7] Photo – Wintersun Chemical](https://cdn.shopify.com/s/files/1/0724/7981/products/19-125-4.jpg?v=1568836921)